Greetings readers! Today, the goal is to update you on some very exciting data that was recently presented at ESMO, the European Society of Medical Oncology Annual Meeting. In a nutshell, early results from a trial using a type of immune therapy called engineered T cells specific for a protein called MAGE-A4 led to significant tumor shrinkage in patients with advanced synovial sarcoma. In this post, I’m going to first translate what I just said… and then lay out my thoughts as to what patients and families need to know about this treatment option, and how it fits in with standard treatment plans and other ongoing clinical trials for synovial sarcoma today. Of NOTE, this study (and trials directed at NY-ESO-1) are also enrolling patients with another rare sarcoma type called myxoid/round cell liposarcoma. While we do NOT yet have any data about results for MAGE-A4 treatment for this sarcoma subtype, there are also early positive results for engineered T cells against a different protein NY-ESO-1 for this disease. So if either of these sarcomas is interesting to you (for any number of reasons) keep reading this article.
Background on synovial sarcoma
Much of this information can be found in more detail in a fantastic review article from Dr. Silvia Stacchiotti and Brian Van Tine.
Synovial sarcoma makes up about 8-10% of all soft tissue sarcomas, and a recent report suggested about 600 new cases were diagnosed in the United States, and about 2100 people living with the disease in 2018. This is a disease of younger adults, most common in ages 20-49. The disease is named based on the appearance of tumor cells under the microscope, which resemble “synovium,” the cells that line joint spaces. The actual cell that these cancers come from is not known, unlike other types of sarcoma (osteosarcoma cells were originally bone, liposarcoma cells come from fat cells, etc.)
Almost all synovial sarcomas develop because of a translocation – this is where two genes that should not be anywhere near each other geographically speaking in the DNA genetic code, get flip-flopped and wind up unhappy neighbors. We don’t understand what causes this, most likely this is purely by chance. The partner genes are named SYT and either SSX1, 2, or 4. Unfortunately this evil duo obtains villainous powers, and leads to cancerous growth, invasion, spread, and other bad behavior. Genetic testing for this partnership is particularly important to confirm the diagnosis of synovial sarcoma, as this is the ONLY disease with these two genes placed together.

Most synovial sarcomas occur in the extremities (arms, legs) although sometimes they can be in other unusual locations (I’ve taken care of a patient with a synovial sarcoma near the prostate gland, several patients with synovial sarcomas beginning in the lung lining or pleura, and some in the abdominal cavity). Synovial can spread to draining glands or lymph nodes (pretty unusual for sarcoma), and often spreads to the lungs as the main site. Overall, synovial sarcoma is quite aggressive, with only 50-60% of adult patients likely to survive for greater than 5 years (historically….) Pediatric patients tend to do better.
Standard treatments for synovial sarcoma
Like other types of sarcoma, treatment for synovial sarcoma that is localized (not spread) revolves around complete surgical removal if possible. Radiation treatment to the site of the sarcoma is often used for large tumors (bigger than 5 cm) or if the edges of the surgery area had cancer under the microscope (positive margins.) Metastatic disease occurs if tumor cells leak out of the main tumor into the blood, and travel to lymph nodes or the lungs or other sites – forming new tumors. Unfortunately we can only find these if they have already started to grow in the other sites on CT scans.
The use of chemotherapy to shrink the cancer before surgery and to kill possible “floaters” is controversial. However multiple studies suggest that chemotherapy, specifically doxorubicin or epirubicin with ifosfamide improves the chances of cure, or at least delays the time before the disease returns. For my synovial patients with large tumors (> 5 cm) and high grade (measure of aggressiveness under the microscope), I typically give between 4 to 6 cycles of doxorubicin/ifosfamide with some before and some after surgery and radiation treatment.
For synovial sarcoma that returns or is metastatic, there are multiple standard treatments that can shrink or slow the growth of the cancer. In some cases the disease can be shrunk enough to allow for surgical removal of the tumors in the lungs. However true cure is very difficult in patients where the disease has spread to other organs which is why we need new treatments. I have had some patients who have had stable disease for months to even years on these treatments. On the other hand, I have had patients where the disease was very aggressive quickly.
Standard Treatments for Metastatic Synovial Sarcoma:
- Doxorubicin with ifosfamide (especially if not used around the time of surgery)
- Ifosfamide alone (standard or high dose)- this is probably the most effective drug to stabilize the disease even in people who have already had it before
- Trabectedin (helps probably 20-25% of patients)
- Pazopanib, a chemo pill that blocks tumor blood vessel growth and signaling. I have had some patients on pazopanib for years with stable or very slowly growing synovial sarcoma. Not sure why this is!
Immune therapy for synovial sarcoma
Now… what about using our immune system to fight sarcoma? Check out my immunotherapy 101 post for a reminder about the basics of this idea. The immune system’s job is to keep an eye on all of our cells, and master immune cells called cytotoxic T cells have the ability to destroy damaged or infected cells. So, all of our normal cells are constantly participating in mandatory self-checks of their proteins by the immune system. Our cells have these “machines” called proteasomes, whose job it is to chop up proteins into little fragments called peptides. On a regular basis, a sampling of the peptide innards of the cell are tacked on to other “reporter” proteins called HLA, and shipped to the surface of the cell like antenna. The cytotoxic T cells check all of these HLA-peptide complexes by matching it to the specific T cell receptor (TCRs). We have thousands and thousands of different shaped TCRs to be able to recognize these HLA-peptide complexes. Now, any T cells with TCRs that can recognize and bind to normal “self” protein particles have been destroyed long ago – we don’t want our immune system to kill off healthy cells with normal protein particles! However, cancer cells DO have “foreign” proteins that can be recognized by the immune system, because the “mutations” or genetic mistakes in cancer lead to new, weirdly-shaped proteins that get chopped up by the proteasomes and presented with HLA just like normal proteins. The best case scenario is that the T cell receptor recognizes the HLA-cancer protein complex, binds tightly, and then activates, killing off the cancer cell. Unfortunately, most cancer proteins resemble self proteins too much – and are not very good at triggering the immune system or binding to TCRs – which is why cancer cells ultimately can avoid being killed off by the immune system.
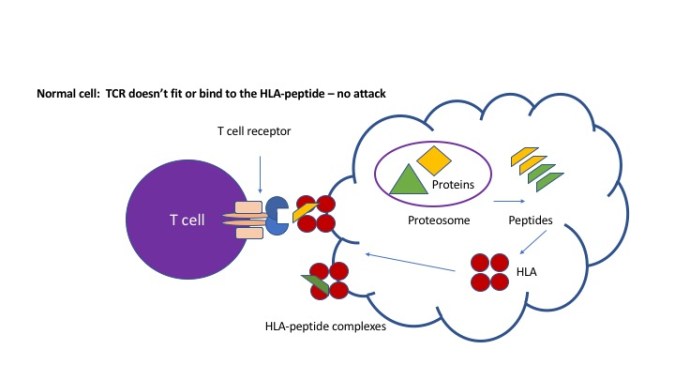
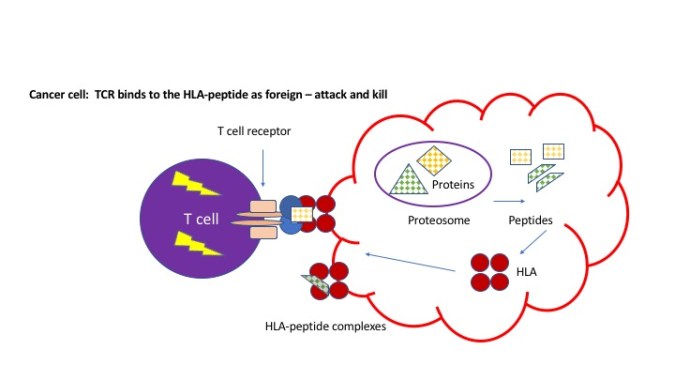
Here is where special proteins called cancer/testis antigens come into play. Cancer/testis antigens are normally ONLY active during development and growth as a fetus in the womb! Appropriately for the goal at the time, they lead to crazy rapid cell growth, differentiation and weird migration patterns – necessary for a growing embryo to turn from a blob of cells to an organized human being. In almost all normal cells these proteins are turned off (silenced) in the DNA once the baby is born. The only place in adults where these antigens are found is in the testes, where sperm growth and maturation happen throughout adulthood. Now, some cancers turn back on these genes, leading to the protein being produced and helping promote growth and spread. So, this is potentially a protein that is unique to the cancer cell that our T cells would not be trained to ignore – so they should be able to attack it as foreign!!! The kicker – and the whole point of this blog) is that about 60% of synovial sarcomas will have cancer/testis antigens –NY-ESO-1 and MAGE-A4 are the two key ones we will discuss now.
This sounds great – but the problem is that most synovial sarcomas have no T cells in the tumors. Meaning, even though they might have a potential “foreign” protein to present, either 1) the T cells are either not binding well enough and strongly enough to the HLA-peptide complex to get activated and kill the synovial sarcoma cells, or 2) there aren’t enough specific T cells produced, or 3) they are getting shut down by other nasty suppressive chemicals and immune cells common in cancer deposits. If you don’t have T cells to begin with that can recognize the tumor, using immune boosting drugs like checkpoint inhibitors (Keytruda, Opdivo, Yervoy, etc) to boost T cell activity doesn’t make much sense. It’s like slamming on the gas pedal when the car is out of gas. You’re not going anywhere.
So researchers beginning with Steven Rosenberg from the National Cancer Institute, and now various drug companies are going after synovial sarcomas that express NY-ESO-1 or MAGE-A4. The ongoing clinical trials are designed to provide patients a super-customized and engineered T cell receptor that binds NY-ESO-1 or MAGE-A4 with the HLA complex very tightly – and stimulates activation and cancer cell kill. The trials are doing this in two different ways:
Engineered T cells:
We collect the synovial sarcoma patients’ own white blood cells including T cells through “pheresis” of the blood, like being hooked up to a blood donating machine. The white blood cells are sorted and grown to pull out T cells in an outside laboratory. But remember, this population of T cells from the blood will have all kinds of different T cell receptors. An inactivated virus is used to deliver the lab-developed supercharged TCR specific for NY-ESO-1 or MAGE-A4 into each of these T cells. These engineered T cells are grown again into a product of about 5-10 billion T cells specific for NY-ESO-1 or MAGE-A4. The product is then sent back to the doctor, the patient receives chemotherapy to clean out other immune cells and make room for the new engineered T cells.
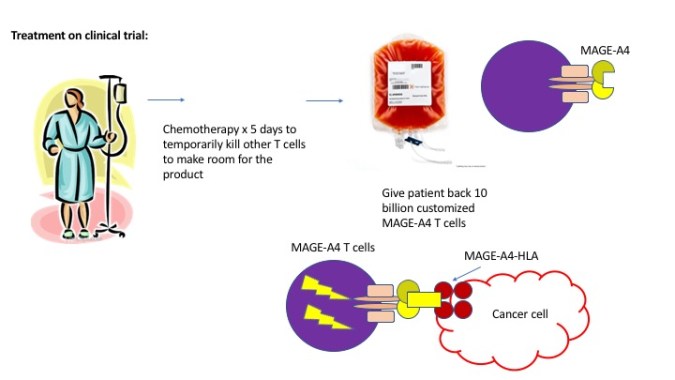
The really cool thing is that these NY-ESO-1 and MAGE-A4 directed T cells look very promising in synovial sarcoma patients. The initial publication and presentation for NY-ESO-1 reported responses (tumor shrinkages) in about 50% of patients, and then the most recent presentation from ESMO for MAGE-A4 showed that 11 out of the 12 patients had stable disease or tumor shrinkage! While the MAGE-A4 data is still very early, patients who had benefit from the NY-ESO-1 treatments could be long-lasting (months to years).
This is not a treatment to be taken lightly though. The doses of conditioning chemotherapy are similar to a bone marrow transplant, so patients will be without a functioning immune system, and might require blood and platelet transfusions for a few weeks. Additionally, after putting in the supercharged T cells all at once, many patients will experience a burst of immune chemicals called cytokines all being dumped at the same time. This is like what happens if you have the flu or a serious other infection – fevers, chills, drop in blood pressure, confusion, organ damage, etc. Fortunately we are used to dealing with this with other immune treatments so we know how to manage it – but this is a big deal.
Soluble T cell receptors:
Other companies have developed something called a “soluble” T cell receptor. This TCR is not attached to a T cell – it’s a free-floating protein all by itself. One end of the TCR has been engineered to bind very tightly to either NY-ESO-1 or MAGE-A4 with the HLA on the tumor cells, and the other end binds to an actual T cell and stimulate activity and tumor kill. It’s a 2-headed monster – of the good kind! We have no data yet for these trials but these are open for synovial sarcoma patients as well as other types of cancer that can express these markers – lung cancer, bladder cancer, and melanoma.
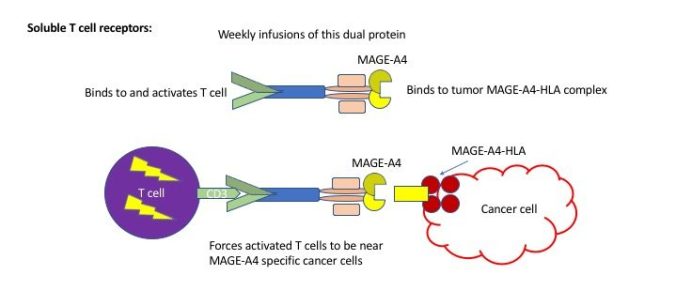
So… what’s the catch? The issue with these types of treatment is that people have many many different HLA types. This is what we have to match between donors to recipients for organ transplants or bone marrow transplants. The different classes of HLAs are shaped differently… so a TCR might be able to recognize the same peptide on one type of HLA but not another. So all of these technologies I was talking about are only going to possibly work (and are only being explored) in people with the HLA type A2. Fortunately, this is about 40% of the population but it varies based on your ethnicity and genetics. So before any patient can participate in these trials, they have to be tested for HLA (a blood test).
So here’s the summary of clinical trial and standard treatment options for patients with metastatic synovial sarcoma heading into 2020.

Selected Specific trials mentioned here (offered at multiple sites): there are other trials that may be options as well.
Engineered T cells for NY-ESO-1 for synovial sarcoma (re-opening soon)
Engineered T cells for NY-ESO-1 for myxoid round cell liposarcoma
Soluble T cell receptors for NY-ESO-1 – synovial sarcoma, lung cancer, bladder cancer, melanoma
Soluble T cell receptors for MAGE-A4 with or without atezolizumab – synovial sarcoma, lung cancer bladder cancer, gastric or esophageal cancer, head and neck cancer, ovarian cancer
Engineered T cells for MAGE-A4 for synovial sarcoma and myxoid-round cell liposarcoma
Anlotinib for synovial sarcoma (and leiomyosarcoma and alveolar soft part sarcoma)
Doxorubicin plus dual immune checkpoint blockade for soft tissue sarcomas
In a nutshell – these immune therapies may or may not be good options for you – it’s important to talk with your doctor and consider the risks of any potential therapy. But the benefits are looking very exciting in patients where other treatments have not been helpful although it’s early. It’s also unfortunate that most patients will not have the right HLA type to receive these therapies – hopefully this will also change in the future with treatments like CAR-T cells for sarcomas. CAR T cells are also patient’s own T cells that are engineered with a supercharged receptor for a specific tumor target, but does not depend on HLA. However we still have a lot of work to do to find the right targets for CAR-T in solid tumors.
Hope this is helpful! I’m super excited to be able to offer these therapies to my own synovial sarcoma patients.
We live in Western Colorado and my husband, Pat Solbach at 70 years old is being treated for myxoid/round cell liposarcoma 10 cm 180 degrees around the popliteal fossa at the sarcoma clinic at Huntsman Cancer Institute, SLC where I received a PBSCT from a HLA match of 10/10 MUD on April 27, 2016 for AML. At his diagnosis after biopsy on July 2, 2019 they assumed the two small lesions on his pleura were the same and the next week started a five week session of doxifos (3 days IV ifosfamide with mesna with first two including doxorubicin push). After each session and the allotted time he received an injection of Nuelasta He skied 48 days last season. He did took the chemo without incident and not much change in the knee according to images but a significant change to range of motion. The pleura lesions reduce about 50%. Three weeks after completing chemo and before starting radiation treatment a biopsy of the pleura lesions confirmed they were myxoid liposarcoma. He’s now had 14 days of the planned 25 days of radiation to both the left chest and right leg. Surgery is planned December 18th.
Due to the fact that stage four usually does not have a cure, we are researching alternatives and were hoping Dr Wilky could help.
LikeLike
Thank you for reaching out. I would certainly consider having him screen for the NY-ESO-1 and MAGE-A4 studies. The nice thing is that he can prescreen, meaning they just test his HLA type and the tumor for the markers, just to find out if he is a candidate or not. He doesn’t have to move forward with getting treatment right away. If you’re interested send me an email and I can direct you to centers that have these trials open where he could screen.
LikeLike
Hi doc,
Thanks for the excellent article. For advanced MRCLS (stage III or IV) are there any promising trials for patients who don’t test positive for HLA-A02? i.e. they have another type?
LikeLike